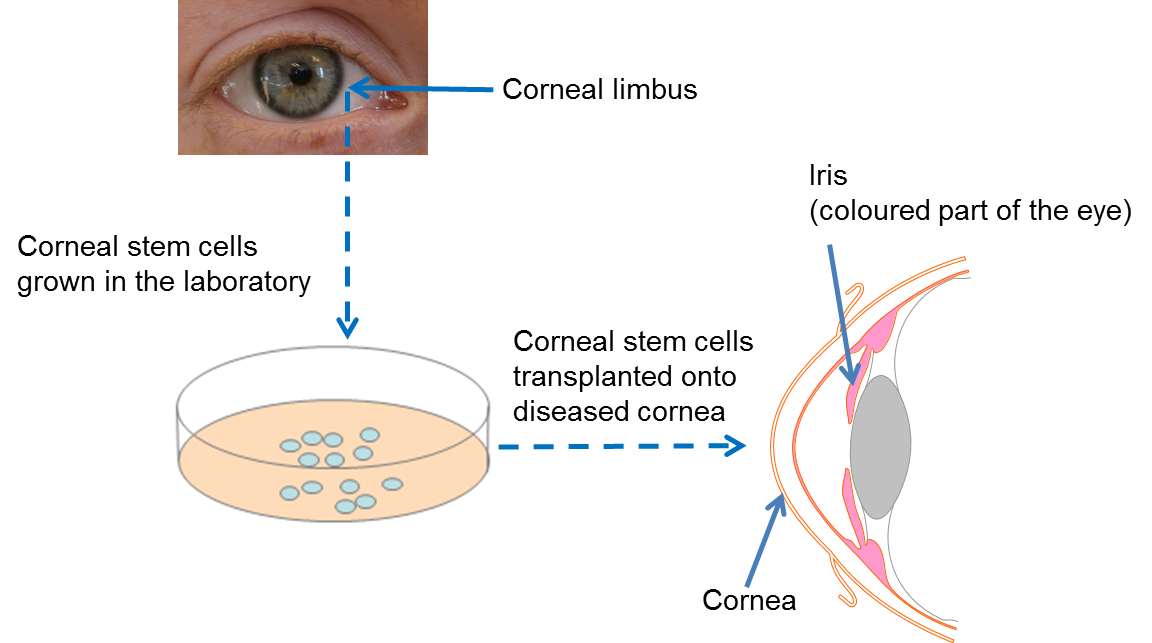
Stem cell therapy for corneal repair is revolutionizing the field of ophthalmology, offering renewed hope for patients suffering from severe corneal injuries. This innovative treatment involves the harvesting of limbal epithelial cells from a healthy eye and their transplantation to the damaged cornea, effectively restoring vision and alleviating pain. Recent clinical trials at Mass Eye and Ear have demonstrated the efficacy of this approach, particularly through the cultivated autologous limbal epithelial cells (CALEC) procedure, which has shown remarkable success rates in restoring corneal surfaces. As a cutting-edge method in corneal injury treatment, stem cell therapies present a viable alternative to traditional approaches, such as corneal transplants, which may not be applicable in cases of significant limbal stem cell deficiency. With ongoing research and potential future advancements, this therapy could transform the landscape of eye damage repair, offering a solution previously deemed unattainable.
The advances in ocular regenerative medicine are paving the way for groundbreaking solutions in vision restoration, particularly through techniques like CALEC surgery. This method, utilizing stem cells to mend corneal damage, represents a significant leap forward in the treatment of eye trauma. By focusing on the regeneration of vital limbal epithelial cells, this therapy addresses the core issue in cases of corneal injury. The implications of such treatments extend beyond mere visual restoration; they promise to alleviate the chronic pain and discomfort associated with severe eye conditions. As research progresses, this eye treatment paradigm could change the lives of countless individuals plagued by vision impairment.
Understanding Stem Cell Therapy for Corneal Repair
Stem cell therapy has emerged as a revolutionary approach for addressing corneal injuries that result in vision loss. Specifically, cultivated autologous limbal epithelial cells, known as CALEC, harness the regenerative properties of limbal stem cells sourced from a healthy eye. This innovative technique has successfully restored the corneal surface in patients who had previously been considered untreatable. The process involves careful extraction of stem cells, cultivation into grafts, and subsequent transplantation, illustrating a significant advancement in the treatment of ocular damage.
The demand for effective solutions like stem cell therapy for corneal repair is underscored by the limitations of traditional treatments, such as corneal transplants. Corneal injuries can arise from various causes, including chemical burns or infections, leading to limbal stem cell deficiency. These deficiencies prevent normal ocular healing and maintenance of eye surface integrity. By utilizing stem cell therapy, clinicians can address these critical issues and improve patient outcomes through successful regeneration of damaged corneal tissues.
The Promise of CALEC Surgery in Corneal Injury Treatment
CALEC surgery represents a significant breakthrough in the realm of corneal injury treatment. This method not only demonstrates a high success rate in restoring corneal integrity but also emphasizes the importance of personalized, patient-specific approaches to eye damage repair. The encouraging results from clinical trials highlight the potential for initiated cellular healing processes, which can greatly benefit patients suffering from corneal scarring or limbal stem cell deficiencies.
Moreover, CALEC surgery is particularly notable for its innovative manufacturing process that allows for the expansion of limbal epithelial cells into a viable graft. This ensures that the graft is both robust and capable of effective integration into the patient’s eye. As the clinical experience and research behind this therapy grow, it faces the prospect of establishing a new standard of care for individuals who have long endured the challenges of corneal injuries that affect their vision.
The Role of Limbal Epithelial Cells in Eye Damage Repair
Limbal epithelial cells play a crucial role in maintaining corneal health and clarity, making them essential to effective eye damage repair. These specialized cells are located at the limbus, the border between the cornea and the conjunctiva, and are responsible for regenerating the corneal epithelium. When these cells are depleted due to injury or disease, the repair mechanisms of the cornea are severely compromised, leading to painful symptoms and visual impairment.
The success of stem cell therapy, particularly CALEC, relies heavily on the ability to harvest and cultivate these vital limbal epithelial cells. By restoring a healthy population of these cells to the corneal surface, patients can experience significant relief from discomfort and a remarkable improvement in vision. This connection illustrates how advancements in stem cell therapies not only facilitate biological recovery but also enhance the quality of life for those affected by corneal conditions.
Safety and Efficacy of Stem Cell Treatment in Clinical Trials
The safety profile of stem cell treatment, particularly CALEC, has been a focal point in ongoing clinical trials. Results from recent studies indicate that the therapy has a strong safety record with minimal adverse events. For instance, while one participant experienced a bacterial infection linked to contact lens wear, the overall outcomes suggest a low risk associated with the procedure itself. These findings are critical, as they lend confidence to both clinicians and patients regarding the viability of stem cell therapies.
In terms of efficacy, the trials have shown remarkable results in restoring corneal surfaces, with success rates reaching beyond 90% in some cases. These positive outcomes reinforce the potential of stem cell therapy as a transformative approach for patients with severe corneal injuries. Continued research is essential to confirm the long-term benefits of CALEC treatment and to expand its applicability across a wider patient demographic.
Future Directions for Stem Cell Therapies in Ophthalmology
As research into stem cell therapies progresses, future studies are expected to explore larger patient populations and more diverse clinical settings. The next phase of trials for CALEC will likely focus on comprehensive follow-up periods to assess the durability of treatment outcomes. This will provide invaluable data to support the long-term effectiveness of stem cell therapy in restoring vision and alleviating corneal trauma.
Another promising avenue for development is the exploration of allogeneic approaches, which involves utilizing limbal stem cells from donor eyes. As discussed by researchers, this strategy could significantly enhance treatment availability, particularly for patients with bilateral corneal injuries. This expansion of eligibility may pave the way for stem cell therapy to become a mainstream treatment option in ophthalmology, revolutionizing how eye damage repair is approached.
Exploring the Clinical Applications of CALEC Transplantation
The clinical applications of CALEC transplantation extend beyond simple corneal surface restoration; it enables a holistic approach to treating complex ocular conditions. With increasing evidence of its effectiveness, CALEC addresses both structural and functional restoration of damaged eyes. This technique signifies a shift toward personalized medicine in ophthalmology, where treatments can be tailored based on individual patient needs.
Additionally, there is an increasing interest in applying CALEC procedures to treat cases that have failed conventional therapies. As more data becomes available, it is likely that CALEC will not only become an alternative solution but also establish itself as a frontline strategy for addressing various corneal pathologies. The potential of stem cell therapy as a game changer in eye care continues to inspire researchers and practitioners alike.
Addressing Regulatory Challenges in Stem Cell Research
While the advancements in stem cell therapies, such as CALEC, are promising, they also face regulatory challenges before becoming widely available. The experimental status of the procedure involves rigorous scrutiny from the U.S. Food and Drug Administration as well as ethics boards, which is essential to ensure patient safety and treatment efficacy. As researchers seek clarity on the approval process, the journey from clinical trial to clinical practice remains critical in the fight against corneal injuries.
Moreover, navigating the complexities of regulatory landscapes can shape the trajectory of future stem cell research. Increased advocacy for funding and streamlined regulatory processes could significantly expedite the availability of these groundbreaking therapies to patients in need. Ensuring comprehensive guidelines and protocols enhances both the safety and feasibility of implementing innovative treatments within mainstream healthcare systems.
Patient Perspectives on Stem Cell Therapy for Corneal Repair
Patents’ experiences with stem cell therapy can provide profound insights into the true impact of CALEC and similar treatments. Testimonials and case studies of individuals who have undergone the procedure often reveal significant improvements in their quality of life, underscoring the emotional and psychological benefits that accompany restored vision. Understanding the patient journey not only highlights the importance of this therapeutic approach but also emphasizes the need for continued research and support.
Additionally, exploring patient perspectives prompts vital conversations about accessibility and the diversifying applications of stem cell therapies. As more patients become aware of such innovative treatments, outreach and education about CALEC and other restorative options become integral. Cultivating awareness in communities can help alleviate fear and misinformation surrounding stem cell applications in eye care, ultimately expanding the reach of these transformative procedures.
The Collaborative Efforts Driving Stem Cell Research Advances
The success of stem cell therapy for corneal repair is a culmination of dedicated collaborative efforts among researchers, medical professionals, and institutions. Partnerships between Mass Eye and Ear and organizations like Dana-Farber Cancer Institute have been instrumental in ensuring robust research that translates into clinical applications. This interdisciplinary approach fosters innovation and accelerates advancements in treatment methodologies for corneal injuries.
Moreover, collaboration extends beyond laboratories and hospitals; public and private funding plays a crucial role in supporting research initiatives. The backing from institutions such as the National Eye Institute highlights the importance of governmental support pathways that facilitate groundbreaking discoveries in ophthalmology. By working together, these entities are shaping the landscape of eye damage repair through stem cell therapies, promising a future filled with potential.
Frequently Asked Questions
What is stem cell therapy for corneal repair and how does it work?
Stem cell therapy for corneal repair involves using cultivated autologous limbal epithelial cells (CALEC) to regenerate damaged corneal surfaces. This innovative treatment extracts healthy stem cells from a donor eye, expands them into a tissue graft in a lab, and then transplants this graft to the injured eye, effectively restoring its surface.
How effective is stem cell therapy eye treatment for corneal injuries?
The stem cell therapy eye treatment known as CALEC has shown over 90% effectiveness in restoring corneal surfaces based on clinical trials. Initial results indicate complete restoration in approximately 50% of patients after three months, with improved success rates seen during follow-up at 12 and 18 months.
What types of corneal injuries can be treated with stem cell therapy for corneal repair?
Stem cell therapy for corneal repair is effective for a variety of corneal injuries, including those caused by chemical burns, infections, and traumatic events that deplete limbal epithelial cells. This therapy is specifically aimed at restoring corneal surfaces when traditional methods, like corneal transplants, are not viable.
What are limbal epithelial cells and their role in eye damage repair?
Limbal epithelial cells are specialized stem cells located at the cornea’s outer margin (the limbus). They play a vital role in maintaining a healthy corneal surface. When damaged by injury or disease, a deficiency of these cells can result in severe visual impairments, making stem cell therapy for corneal repair essential for recovery.
Are there any risks associated with stem cell therapy for corneal repair?
While stem cell therapy for corneal repair has a strong safety profile, minor risks can include infections or complications related to the transplant procedure. In clinical trials, there were few adverse events, with one instance of a bacterial infection that was manageable. Overall, the procedure has proven to be safe for the majority of participants.
How long does the stem cell therapy eye treatment process take?
The stem cell therapy eye treatment process typically involves a two to three-week period for laboratory expansion of the extracted limbal epithelial cells into a graft, followed by the surgical transplant into the damaged eye. Thus, patients can expect the total time from biopsy to graft surgery to be several weeks.
Is stem cell therapy for corneal repair available to the general public?
Currently, stem cell therapy for corneal repair, specifically through CALEC surgeries, remains experimental and is not widely available to the general public. Further studies and clinical trials are necessary before the procedure can receive federal approval for routine administration.
What advancements have been made in stem cell therapy for corneal repair?
Significant advancements in stem cell therapy for corneal repair include the successful completion of clinical trials that demonstrated the safety and effectiveness of CALEC. As a result, researchers aim to expand treatment capabilities, potentially allowing for the processing of donor stem cells for patients with bilateral eye damage in the future.
How can patients participate in future studies on stem cell therapy for corneal repair?
Patients interested in participating in future studies on stem cell therapy for corneal repair should consult their eye care providers or contact research hospitals like Mass Eye and Ear, where new trials may be announced. Participation typically requires meeting specific eligibility criteria.
| Key Points |
|---|
| Mass Eye and Ear conducts the first CALEC surgery for corneal repair. |
| Stem cell therapy from healthy eyes restores corneal surfaces in patients. |
| 14 patients treated in a clinical trial with monitoring lasting 18 months. |
| CALET demonstrated over 90% effectiveness in restoring corneal surfaces. |
| This procedure involves harvesting, expanding, and transplanting stem cells. |
| Success rates for complete corneal restoration increased over 18 months. |
| Overall trial results indicated high safety with minimal adverse effects. |
| Further studies are necessary for FDA approval and larger patient trials. |
Summary
Stem cell therapy for corneal repair is a groundbreaking advancement in ophthalmology, providing hope for patients with blinding corneal injuries. Implemented through cultivated autologous limbal epithelial cells (CALEC), this innovative treatment has shown over 90% efficacy in restoring corneal surfaces among clinical trial participants. The procedure not only highlights the potential for regenerative medicine in eye care but also sets a foundation for future advancements in stem cell applications, promising wider availability and improved outcomes for patients suffering from severe corneal damage.